In the field of modern medical device manufacturing, Injection Molding has become one of the key processes for manufacturing core components of medical devices, relying on its core advantages of high precision, high efficiency, high consistency, and scalable production. Medical devices have extremely strict requirements on the biocompatibility, dimensional accuracy, sterility, corrosion resistance, and structural stability of components. Through precise process control, adaptation of special medical materials, and refined mold design, injection molding technology can perfectly meet these core needs and is widely used in various sub-fields such as disposable medical consumables, implantable medical devices, and diagnostic equipment components. Combining typical domestic and foreign application cases, this paper will elaborate on the specific applications, technical difficulty breakthroughs, implementation processes, and application effects of injection molding technology in medical device manufacturing, demonstrating its important role in promoting the development of medical devices towards miniaturization, precision, intelligence, and low cost.
I. Core Advantages and Application Premises of Injection Molding Technology in Medical Device Manufacturing
The core difference between medical device manufacturing and ordinary industrial product manufacturing is that its products are directly related to patients' life safety, so there are nearly harsh standards for production processes and product quality. The reason why injection molding technology can become the mainstream process in medical device manufacturing lies in its irreplaceable advantages: first, high-precision molding capability, which can achieve micron-level dimensional tolerance control to meet the precision structural requirements of medical device components (such as catheters and implants); second, strong material adaptability, which is compatible with various medical-grade polymer materials, including polycarbonate (PC), polypropylene (PP), polyetheretherketone (PEEK), liquid silicone rubber (LSR), etc., and can ensure that the biocompatibility of materials is not damaged through process optimization; third, high production efficiency, batch production can be realized with multi-cavity molds, and the molding time per mold is short, which can meet the large-scale supply demand of disposable medical consumables; fourth, flexible structural molding, which can realize integrated molding of complex cavities, thin walls, and multi-components, reducing subsequent assembly processes and pollution risks; fifth, controllable cost, large-scale production can significantly reduce the unit product cost, while reducing material waste, which is in line with the industry trend of improving the cost performance of medical devices.
At the same time, the application of injection molding technology in medical device manufacturing must meet strict prerequisites: first, the materials must pass biocompatibility certification and comply with the ISO 10993 series standards to ensure no cytotoxicity, no sensitization, and no teratogenicity. Especially for components that are in direct contact with human blood and tissues, strict biological testing is required; second, the production environment must meet the corresponding cleanliness level, usually requiring an ISO Class 7 (10,000-class) or higher clean workshop to avoid dust and microbial pollution during the production process; third, a complete quality control system must be established in the process to realize full-process traceability from raw material procurement, mold processing, injection molding to finished product inspection, complying with the ISO 13485 medical device quality management system requirements; fourth, mold design must balance precision and practicality, using corrosion-resistant and easy-to-clean mold materials to ensure mold service life and product consistency.
II. Typical Application Cases of Injection Molding in Medical Device Manufacturing
Case 1: Application of Two-Shot Molding in Hemostatic Valve Manufacturing
1. Project Background and Requirements
Hemostatic valves are indispensable core components in interventional therapy, mainly used for sealing vascular interventional catheters to prevent blood leakage during surgery and ensure the safety and effectiveness of minimally invasive surgery. The first-generation hemostatic valve previously produced by an international medical device OEM enterprise adopted the method of separate molding of multiple components followed by manual assembly, which had many drawbacks: first, the assembly process was cumbersome, increasing labor costs and production cycles, and manual operation was prone to poor component fitting; second, the product scrap rate was too high, and component damage and poor sealing were likely to occur during assembly; third, it could not meet the arterial pressure requirements, and the tactile feel of the valve was poor, affecting the doctor's operation experience; fourth, the multi-component structure led to a complex supply chain and increased difficulty in compliance verification. To solve the above problems, the enterprise collaborated with Freudenberg Medical to upgrade and optimize the hemostatic valve using two-shot injection molding technology, achieving dual improvement in product performance and production efficiency.
2. Injection Molding Scheme Design and Implementation
The core difficulty of this project was to realize the integrated molding of the hemostatic valve while meeting the requirements of sealing performance, pressure resistance, and tactile feel. The specific implementation process is as follows:
(1) Material Selection: Combined with the functional requirements of the hemostatic valve, two liquid silicone rubbers (LSR) with different Shore hardness were used as molding materials. The combination of hard silicone and soft silicone not only ensures the structural stability of the valve but also provides good flexibility and sealing performance, ensuring that it can withstand arterial pressure and improve the operational tactile feel. The selected silicone materials have all passed ISO 10993 biocompatibility certification, are non-cytotoxic, can be in direct contact with blood, and have excellent chemical resistance and sterilization stability, which can adapt to both ethylene oxide (EO) sterilization and radiation sterilization.
(2) Mold Design: A two-shot injection mold was adopted, integrating two independent barrels and nozzles, which can inject two silicone materials with different hardness respectively. The mold was made of S136 stainless steel, and its hardness reached HRC 50-55 after nitriding treatment, with excellent corrosion resistance and wear resistance. At the same time, it was processed by ultra-precision five-axis equipment, with a positioning accuracy of ±0.001mm. The mold runner was optimized by CAE software to control the pressure difference of the complex cavity ≤5MPa, avoiding defects such as insufficient filling and bubbles; at the same time, a special exhaust structure was designed to prevent gas retention from causing product molding defects. In addition, the mold integrated the end cover and pressure cover of the hemostatic valve into an integral cavity to realize integrated molding, completely eliminating the subsequent manual assembly process.
(3) Process Parameter Optimization: The key to two-shot injection molding is to control the injection sequence and fusion effect of the two materials to avoid delamination and debonding. After many tests, the optimal process parameters were determined: the first shot injected hard silicone, the barrel temperature was controlled at 120-130℃, the injection pressure was 80-100MPa, the injection speed was controlled in multiple stages, the first stage speed was 60mm/s, and the last stage was decelerated to 20mm/s to ensure uniform filling and shaping of the hard silicone in the cavity; the second shot injected soft silicone when the hard silicone was not fully cured, the barrel temperature was 110-120℃, the injection pressure was 60-80MPa, the injection speed was 40-50mm/s, and the two silicones were closely combined without delamination by precisely controlling the holding pressure (decreasing in three stages from 70MPa to 30MPa) and holding time. The cooling time was dynamically adjusted according to the product wall thickness to ensure that the mold cavity pressure dropped below 0.5MPa before mold opening to avoid product deformation.
(4) Production Environment and Quality Control: The production workshop adopted ISO Class 7 cleanliness standard, equipped with a vertical unidirectional flow purification system, with an air change rate ≥50 times per hour, ensuring that the suspended particle concentration ≤352,000 particles/m³ (≥0.5μm). Operators were required to wear full dust-free clothing, and work after dust removal in an air shower and hand disinfection; the injection molding machine adopted a fully enclosed structure, equipped with a HEPA filtration system to prevent oil mist pollution. In terms of quality control, samples of each batch of products were tested for dimensional accuracy (using a coordinate measuring machine, the tolerance compliance rate reached 99.97%), sealing performance (ensuring no leakage under arterial pressure), biocompatibility, and appearance inspection. A full-process traceability system was established to record information such as raw material batches, process parameters, and inspection results.
3. Application Effects
Through the application of two-shot injection molding technology, the hemostatic valve product has achieved significant upgrades: first, the product performance has been greatly improved. The combination of hard silicone and soft silicone meets the arterial pressure requirements, has excellent sealing performance, can effectively reduce patients' blood loss during minimally invasive surgery, and the good tactile feel improves the doctor's operational convenience; second, the production efficiency has increased by more than 40%. After eliminating the manual assembly process, the molding time per mold is shortened to less than 30 seconds, and the scrap rate is reduced from 15% to less than 2%; third, the cost is significantly reduced, labor costs are reduced by 60%, material waste is reduced by 30%, and the supply chain and compliance verification process are simplified; fourth, the product consistency is greatly improved, and the dimensional tolerance is controlled within ±0.01mm, ensuring the uniform performance of each product. At present, this hemostatic valve has been widely used in cardiovascular, peripheral vascular, and neurovascular interventional surgeries, and has been recognized by many medical institutions around the world.
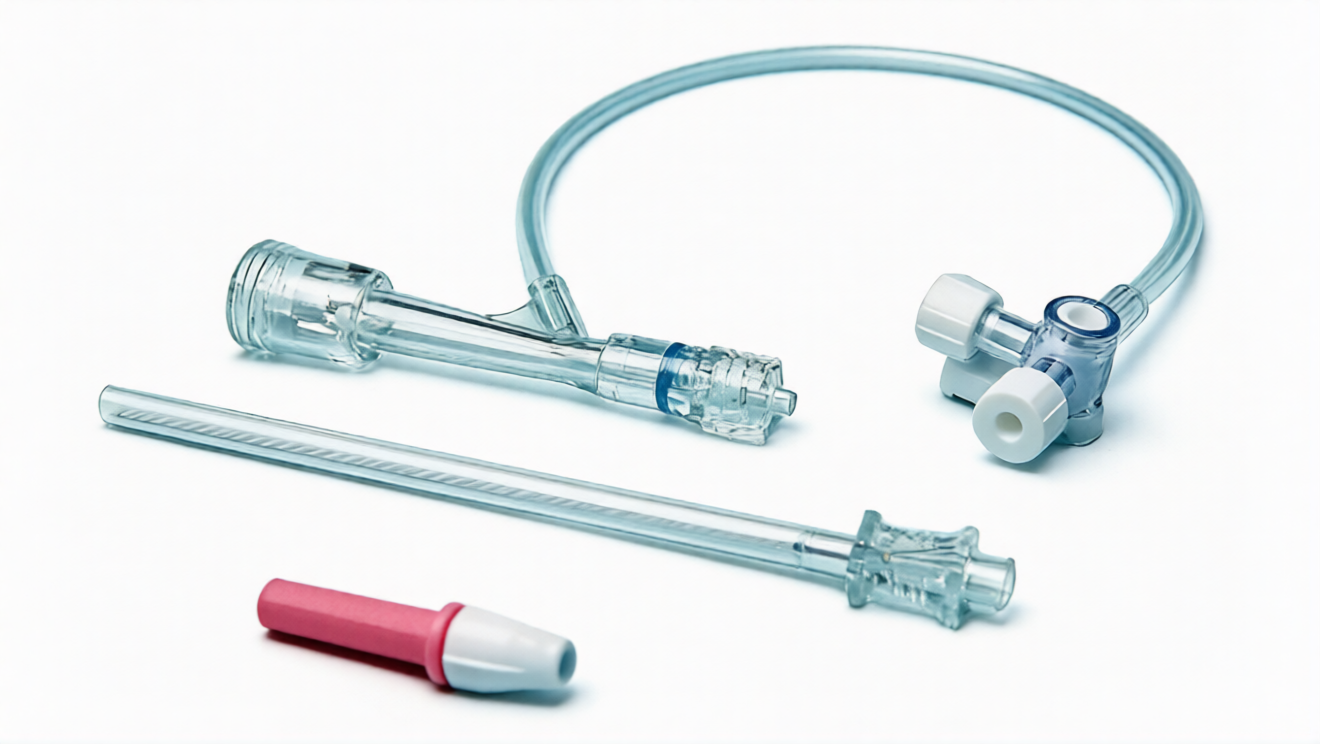
Case 2: Application of Precision Injection Molding in Auto-Injector Housing Manufacturing
1. Project Background and Requirements
Auto-injectors are drug delivery devices that can automatically provide injection doses for patients. In recent years, they have been widely used for self-injection of drugs such as epinephrine and insulin. Their core requirements are miniaturization, portability, low cost, and high reliability. Pirouette Medical, an American company, planned to develop a patented auto-injector, requiring the product to be small in size and light in weight for easy carrying by patients, while having excellent structural strength and durability to withstand the pressing impact force during injection, and the manufacturing cost to be lower than that of existing similar products. Since the auto-injector housing needs to have transparency (to facilitate observing the remaining amount of medicine), high toughness, and dimensional stability, traditional manufacturing processes are difficult to meet the requirements. Therefore, the company chose to cooperate with Covestro to produce housing components using precision injection molding technology.
2. Injection Molding Scheme Design and Implementation
The core of this project was to select suitable medical-grade materials and realize the molding of thin-walled, miniaturized housings through precision injection molding processes. The specific implementation process is as follows:
(1) Material Selection: After multiple rounds of screening, Covestro Makrolon® 2458 medical-grade polycarbonate (PC) was finally selected as the molding material. This material has excellent biocompatibility, complies with ISO 10993-1 standard, and is non-cytotoxic and non-sensitizing; it also has high fluidity, high toughness, and good dimensional stability, which can realize thin-walled design (the thinnest wall thickness is only 0.8mm) to meet the miniaturization and portability requirements of auto-injectors; in addition, this material has strong injection molding capability, low viscosity, is easy to demold after processing, can effectively improve production efficiency, and supports transparent and opaque color customization to meet product appearance design requirements.
(2) Mold Design: The mold adopted ultra-precision processing technology, selected SKD61 mold steel, and had excellent wear resistance and corrosion resistance after nitriding treatment. The mold cavity was designed according to the complex structure of the auto-injector housing, and a hot runner system was used to optimize the runner layout, reduce weld lines, and avoid stress concentration; for the thin-walled structure, a draft angle of 3°-5° was designed to control the product deformation ≤0.2mm; at the same time, a vacuum exhaust groove was set to prevent gas retention from forming bubble defects. The mold adopted a multi-cavity design (8 cavities), which can realize batch production and greatly improve production efficiency. In addition, the mold was equipped with a conformal waterway cooling system, which improved the mold temperature uniformity by 40% and reduced product deformation caused by thermal stress.
(3) Process Parameter Optimization: A precise process parameter matrix was established according to the characteristics of PC materials. The barrel temperature was controlled in sections: the front section temperature was 280-290℃, the middle section was 300-310℃, the rear section was 310-320℃, and the nozzle temperature was 5-10℃ lower than the rear section to avoid material degradation; the injection speed adopted multi-stage injection, the first stage speed was 80mm/s (fast filling the cavity), the middle stage was decelerated to 50mm/s (avoiding melt turbulence), and the last stage was decelerated to 20mm/s (filling the end of the cavity to reduce internal stress); the holding pressure decreased in three stages from 80MPa to 30MPa, and the holding time was 15-20 seconds to ensure full shaping of the product; the cooling time was adjusted to 20-25 seconds according to the product wall thickness, and a water chiller was used to precisely control the cooling water temperature to ensure uniform mold temperature.
(4) Quality Control: A full-process quality control system was established. When purchasing raw materials, the supplier's qualifications were verified, requiring FDA 510(k) certification, CE mark, and complete biocompatibility test report; each batch of materials was sampled to test parameters such as melt flow index and shrinkage rate, with the fluctuation range controlled within ±5%. During the injection molding process, an infrared temperature measurement system and a mold cavity pressure sensor were deployed to collect more than 200 process parameters in real time, and the parameter fluctuation was monitored through Statistical Process Control (SPC) to ensure that the process capability index Cpk ≥1.33. In terms of finished product inspection, Automated Optical Inspection (AOI) was used to check product appearance defects (such as scratches, bubbles, and flash), a coordinate measuring machine was used to detect key dimensions, and tensile strength and impact toughness tests were carried out to ensure that the product met the design requirements.
3. Application Effects
The application of precision injection molding technology has successfully helped Pirouette Medical achieve the design goals of the auto-injector: first, the product has achieved miniaturization and portability, the housing wall thickness is controlled between 0.8-1.2mm, and the product weight is 30% lighter than similar products, which is convenient for patients to carry and use; second, the product has excellent performance, the high toughness of Makrolon® 2458 material ensures that the housing can withstand the pressing impact force during injection without fracture or deformation, has good dimensional stability, and the service life can reach more than 5 years; third, the production efficiency has been greatly improved, the molding time per mold of the 8-cavity mold is only 25 seconds, the daily output can reach more than 10,000 pieces, and the unit product manufacturing cost is reduced by 25%; fourth, the product meets global medical compliance standards, has successfully passed FDA 510(k) and CE certification, and has been promoted and used in many countries and regions around the world. This case fully reflects the core role of precision injection molding technology in the miniaturization and low-cost manufacturing of medical drug delivery devices.
Case 3: Application of IML Injection Molding in Medical Centrifuge Tube Manufacturing
1. Project Background and Requirements
Centrifuge tubes are commonly used medical consumables in laboratory diagnosis and clinical testing, mainly used for sample storage, centrifugal separation and other operations. Their core requirements are precise dimensions, good sealing, clear labels, and easy recycling. Traditional centrifuge tubes adopt the method of pasting labels after injection molding, which has problems such as easy label falling off, inaccurate positioning, and difficult recycling. In addition, the manual labeling process is cumbersome, increasing production costs and pollution risks. To solve the above problems, ARBURG adopted IML (In-Mold Labeling) injection molding technology to produce integrated labeled medical centrifuge tubes, meeting the high-precision and high-reliability requirements of laboratory diagnosis, and improving the sustainability of products.
2. Injection Molding Scheme Design and Implementation
The core of this project was to realize the integrated molding of centrifuge tubes and labels, ensure accurate label positioning and non-falling, and realize product recyclability. The specific implementation process is as follows:
(1) Material Selection: Both the centrifuge tube body and the label were made of medical-grade polypropylene (PP) material, realizing a single material composition, which greatly simplified the recycling process and met environmental protection requirements. The selected PP material has excellent biocompatibility, complies with ISO 10993 standard, is non-cytotoxic, and can be in direct contact with biological samples; it also has good chemical resistance, can withstand a variety of laboratory reagents, and has excellent heat resistance, which can adapt to high-temperature sterilization treatment. The label was made of ultra-thin PP material with scratch-resistant characteristics, which can print precise liquid level scales, temperature monitoring marks, and QR codes containing recycling and warehousing information, facilitating sample traceability and operation.
(2) Equipment and Mold Configuration: ARBURG ALLROUNDER 520 A Ultimate electric injection molding machine was adopted. This equipment has a clamping force of 1500kN, equipped with a servo electric drive device, which can realize precise positioning of tools and high repeatability of the injection process, ensuring the precision and consistency of injection molding. At the same time, it integrated components from three companies: Beck Automation, Kebo, and Intravis to build a one-stop turnkey system to realize automatic in-mold labeling operation. The mold adopted a multi-cavity design, combined with IML technology, to complete label lamination synchronously during the injection molding process. The spacing between labels was controlled within 0.2mm, which significantly reduced quality errors and the number of scrap products. The mold was made of S136 stainless steel, processed by ultra-precision to ensure the smooth surface of the cavity, which is easy to demold and clean.
(3) Process Parameter Optimization: The key to IML injection molding is to control the positioning accuracy of the label and its fit with the tube body to avoid label offset, bubbles, debonding and other problems. The determined optimal process parameters are as follows: the barrel temperature was controlled at 200-240℃, the injection pressure was 70-90MPa, and the injection speed was controlled in multiple stages to ensure uniform filling of the cavity with the melt and avoid label displacement caused by melt impact; the holding pressure was 50-70MPa, and the holding time was 10-15 seconds to ensure close fit between the label and the tube body; the cooling time was 15-20 seconds, and conformal waterway cooling was adopted to ensure rapid product shaping and reduce deformation. In addition, the label needs to be electrostatically treated before being put into the mold to enhance the adsorption force with the melt and ensure firm bonding.
(4) Clean Production and Quality Control: The production workshop met the ISO Class 7 cleanliness standard, and both the injection molding machine and the automation system were covered with air purification modules, including ionization devices, meeting the ISO 13485 standard and FDA regulations. In terms of quality control, samples of each batch of products were tested for dimensional accuracy (key dimensional tolerance controlled within ±0.01mm), label positioning (offset ≤0.1mm), sealing performance (ensuring no leakage), and appearance inspection. At the same time, the wear resistance and chemical resistance of the label were tested to ensure that the label was clearly distinguishable during use. A full-process traceability system was established to realize the traceability of raw materials, process parameters, and inspection results.
3. Application Effects
The application of IML injection molding technology has completely solved the pain points of traditional centrifuge tubes: first, it has realized the integrated molding of centrifuge tubes and labels, with accurate label positioning (offset ≤0.1mm) and firm bonding, which is not easy to fall off, avoiding errors and pollution risks caused by manual labeling; second, the product recyclability has been greatly improved. Both the centrifuge tube body and the label are made of PP material, and the single material structure simplifies the recycling process, which is in line with the environmental protection trend; third, the production efficiency has increased by more than 50%. The automatic IML process has eliminated the manual labeling process, the molding time per mold is shortened to less than 20 seconds, and the scrap rate is reduced to less than 1%; fourth, the product function has been optimized. The scratch-resistant label has precise and clear scales, which is convenient for operators to observe the sample liquid level. The QR code can realize additional functions such as sample traceability and warehousing management. In the future, RFID barcodes can be integrated to realize comprehensive tracking of processes, quality, and patient data. At present, this IML centrifuge tube has been widely used in major laboratories and medical institutions around the world, becoming a benchmark product in the field of medical diagnostic consumables.

Case 4: Application of Ultra-Thin Wall Precision Injection Molding in Insulin Pump Housing Manufacturing
1. Project Background and Requirements
Insulin pumps are core equipment for the treatment of type 1 diabetes. Their housings need to have the characteristics of ultra-thin, light weight, uniform light transmission, and good sealing. They must be easy to carry by patients (almost no sense of weight), protect internal precision electronic components and medicines, and ensure uniform light transmission to facilitate observing the remaining amount of medicines. Hisense Medical once faced an urgent order to produce 100,000 insulin pump housings, requiring a wall thickness of only 0.12mm, a light transmission uniformity error of no more than 3%, and the drug residue per housing must be less than 0.001 milliliters. Traditional injection molding processes could not meet such strict requirements, and German suppliers quoted high prices with long delivery cycles. To this end, Fuying Medical collaborated with Hisense Medical to adopt ultra-thin wall precision injection molding technology to overcome technical difficulties and complete the order delivery.
2. Injection Molding Scheme Design and Implementation
The core difficulty of this project was to realize the precise molding of 0.12mm ultra-thin walls, while controlling the light transmission uniformity and drug residue. The specific implementation process is as follows:
(1) Material Selection: Medical-grade polycarbonate (PC) was selected as the molding material. This material has excellent transparency, high toughness, and dimensional stability, which can realize ultra-thin wall molding. It also complies with ISO 10993 biocompatibility certification, is non-cytotoxic, and can be in direct contact with medicines. To solve the nano-level oriented crystallization problem (affecting drug adhesion) in ultra-thin wall injection molding, the PC material was modified by adding a special anti-crystallization agent to ensure uniform crystallization of the material during the molding process and reduce drug residue.
(2) Mold Design: An ultra-precision nano-coating mold was adopted, with the mold surface roughness reduced to Ra0.012, which reduced the melt flow resistance, avoided flow marks, and improved product light transmission; the mold cavity was optimized by CAE software, and the runner adopted a micro-runner structure to ensure uniform filling of the melt in the ultra-thin cavity; it was equipped with a conformal waterway cooling system, and the mold temperature control accuracy reached ±0.3℃, far higher than the national standard of ±5℃, ensuring uniform product cooling and reducing deformation caused by thermal stress; a special demolding mechanism was designed, and for the ultra-thin wall structure, a flexible demolding method was adopted to avoid product damage during demolding.
(3) Process Parameter Optimization: Dual servo closed-loop control technology was adopted to synchronously adjust the injection speed and mold cavity pressure within 0.02 seconds, ensuring that the temperature difference of the melt flow front in the mold cavity did not exceed 2 degrees Celsius, avoiding invisible flow marks; an adaptive holding pressure algorithm was developed to allow the process parameters of each molding cycle to be automatically fine-tuned according to the ambient temperature and humidity, ensuring product consistency; the barrel temperature was controlled at 290-320℃, the injection speed was controlled in multiple stages, the first stage speed was 100mm/s (fast filling the ultra-thin cavity), and the last stage was decelerated to 30mm/s (avoiding melt impact on the cavity); the holding pressure decreased in four stages from 90MPa to 20MPa, and the holding time was 8-10 seconds to ensure full shaping of the product; the cooling time was 12-15 seconds, and a low-temperature water chiller was used to precisely control the cooling water temperature to ensure rapid product shaping and uniform light transmission.
(4) Production Environment and Quality Control: The production workshop adopted ISO Class 7 cleanliness standard, equipped with a high-precision environmental control system to monitor parameters such as temperature, humidity, and suspended particle concentration in real time, avoiding environmental factors affecting product quality. In terms of quality control, a laser thickness gauge was used to randomly sample each batch of products, with the wall thickness fluctuation controlled within 0.003mm; professional light transmittance testing equipment was used to ensure that the light transmission uniformity error did not exceed 3%; liquid chromatography-mass spectrometry (LC-MS) was used to detect the drug residue on the product surface to ensure it was less than 0.001 milliliters; at the same time, sealing tests and impact toughness tests were carried out to ensure that the product could withstand collisions and extrusions in daily use and protect internal components.
3. Application Effects
The successful application of ultra-thin wall precision injection molding technology not only completed the urgent order delivery smoothly but also achieved multiple technological breakthroughs: first, the product performance reached the international leading level, the wall thickness was controlled at 0.12mm, the wall thickness fluctuation ≤0.003mm, the light transmission uniformity error ≤2.5%, and the drug residue ≤0.0008 milliliters, all better than the customer's requirements, even exceeding the standards of similar German products; second, the product experience was greatly improved. The ultra-thin wall design reduced the weight of the insulin pump housing by 40%, and patients could hardly feel the weight when wearing it, which was especially suitable for pediatric patients, helping diabetic children better integrate into normal life; third, the production efficiency was significantly improved, the molding time per mold was shortened to 18 seconds, the daily output could reach more than 8,000 pieces, the unit product cost was reduced by 30%, and compared with German suppliers, the customer's procurement cost was greatly reduced; fourth, a technical barrier was formed, mastering the core technology of ultra-thin wall precision injection molding, laying a foundation for the production of high-end medical device components in the future. At present, this insulin pump housing has been mass-produced and applied to Hisense Medical's insulin pump products, winning wide market recognition.
III. Application Trends and Challenges of Injection Molding Technology in Medical Device Manufacturing
1. Application Trends
With the development of medical devices towards miniaturization, precision, intelligence, and personalization, the application of injection molding technology in medical device manufacturing will show the following trends: first, multi-material integrated molding will become the mainstream, and two-shot, multi-shot injection molding technologies will be widely used in the manufacturing of complex medical components to realize the combination of materials with different properties and improve product functions; second, intelligent injection molding will become the development direction, and through the introduction of AI, Internet of Things and other technologies, real-time monitoring, automatic adjustment, and predictive maintenance of injection molding process parameters will be realized to improve production efficiency and product consistency; third, the application of micro-injection molding technology will continue to expand. For micro-medical devices (such as micro-catheters and implantable sensors), micro-injection molding technology will realize nano-level precision molding to meet the miniaturization and minimally invasive medical needs; fourth, environmentally friendly development, using degradable medical polymer materials, combined with injection molding technology, to produce degradable medical consumables and reduce medical waste pollution; fifth, accelerated process integration, injection molding technology will be integrated with 3D printing, micro-nano manufacturing and other technologies to realize rapid manufacturing of personalized medical devices and meet the personalized treatment needs of patients.
2. Facing Challenges
Although injection molding technology is widely used in medical device manufacturing, it still faces some challenges: first, the dependence on imported high-end medical materials. The production technology of domestic medical-grade special engineering plastics (such as PEEK, LSR) still needs to be improved, and some high-end materials need to be imported from abroad, increasing production costs and supply chain risks; second, the high requirements for mold processing precision. The mold design and processing of complex medical components are difficult, putting forward higher requirements for the technical level of mold manufacturing enterprises; third, the difficulty of quality control. Medical device components have strict requirements on biocompatibility, sterility, dimensional accuracy, etc., and full-process quality control requires a lot of manpower, material resources, and financial resources; fourth, the increasing compliance requirements. Countries around the world have increasingly strict supervision on medical devices, and the injection molding process needs to comply with multiple standards such as ISO 13485, FDA 510(k), and MDR, resulting in high compliance verification costs and long cycles.
IV. Conclusion
As a core process in medical device manufacturing, Injection Molding technology has been widely used in disposable medical consumables, implantable medical devices, diagnostic equipment components and other fields, relying on its advantages of high precision, high efficiency, high consistency, and strong material adaptability. The four typical cases introduced in this paper—hemostatic valves, auto-injector housings, medical centrifuge tubes, and insulin pump housings—fully demonstrate the important role of injection molding technology in solving the pain points of medical device manufacturing, improving product performance, reducing production costs, and increasing production efficiency.
With the continuous progress of medical technology and the continuous upgrading of market demand, injection molding technology will continue to innovate, gradually developing towards multi-material, intelligent, micro-nano, and environmentally friendly directions, and will also face challenges in materials, molds, quality control, and compliance. In the future, enterprises and scientific research institutions need to strengthen cooperation, overcome core technologies, improve the localization level of medical materials and molds, improve the quality control system, and promote the in-depth application of injection molding technology in medical device manufacturing, providing more reliable, efficient, and cost-effective manufacturing solutions for the development of global medical and health undertakings, and guarding life and health with precision manufacturing.
Hot Articles
Hot Tags
Manufacturing on Demand
Please fill in the following information to obtain plan details (information is confidential and not disclosed publicly), we will contact you within 24 hours, please keep your phone available!
Upload a 3D/2D model to see instant pricing, lead time, and DFM feedback.








